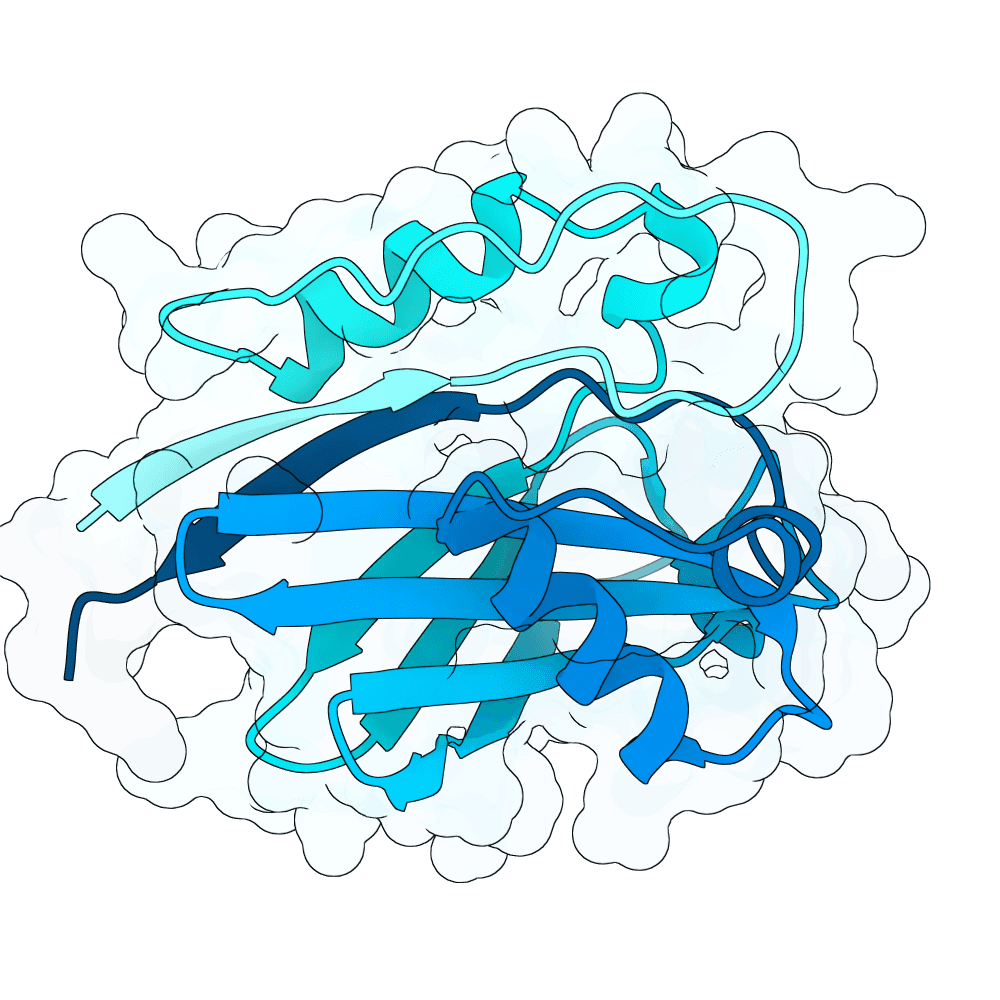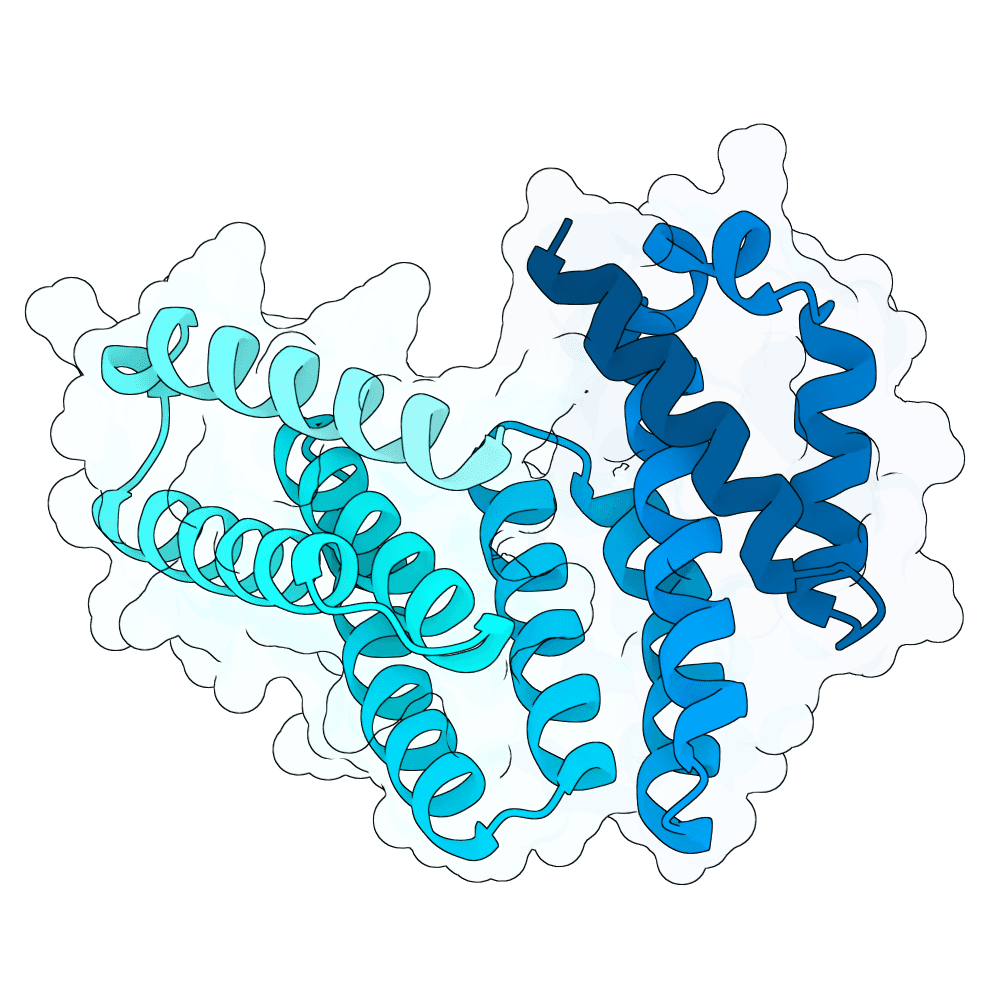Description
Docs: https://docs.google.com/document/d/1GDRoRjBD0ItnyppZOGs8NB8i7ZfTNA5oSimHzEs65UY
Leonardo V. Castorina & Florian Wuennemann
- Introduction
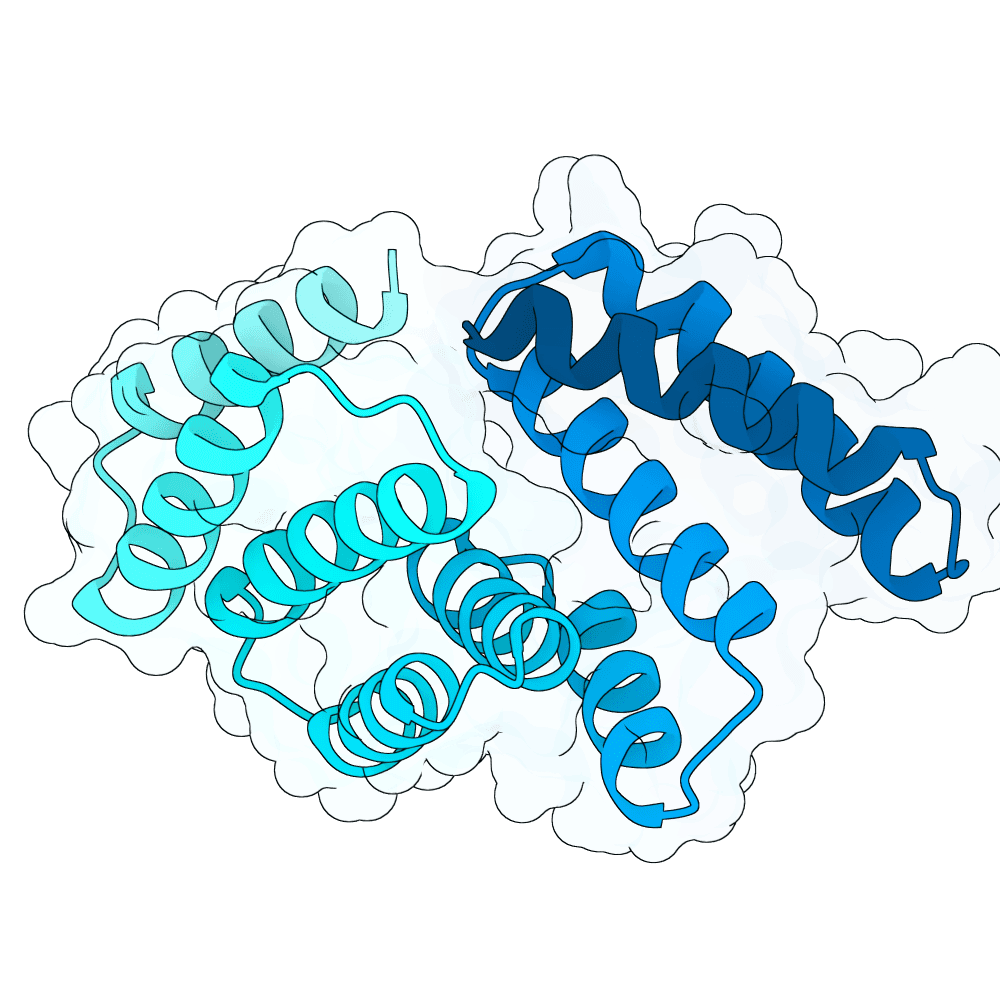
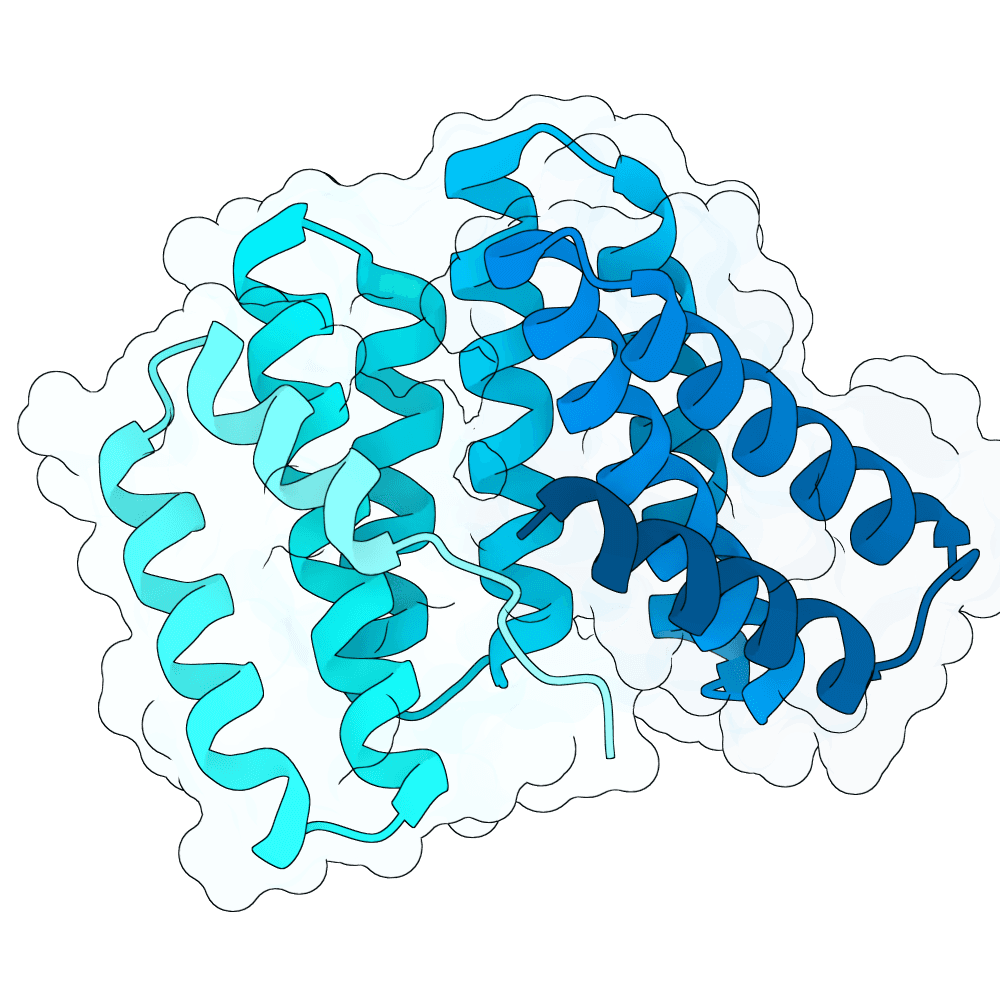
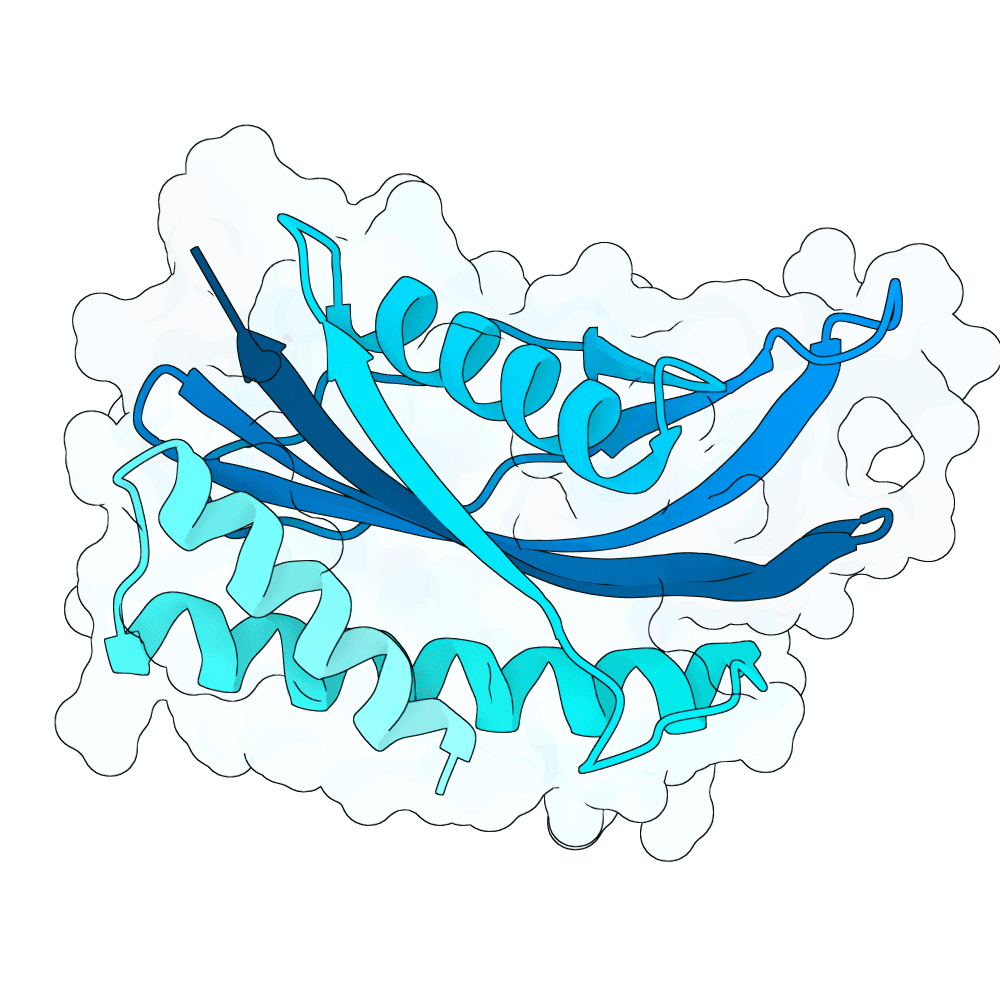
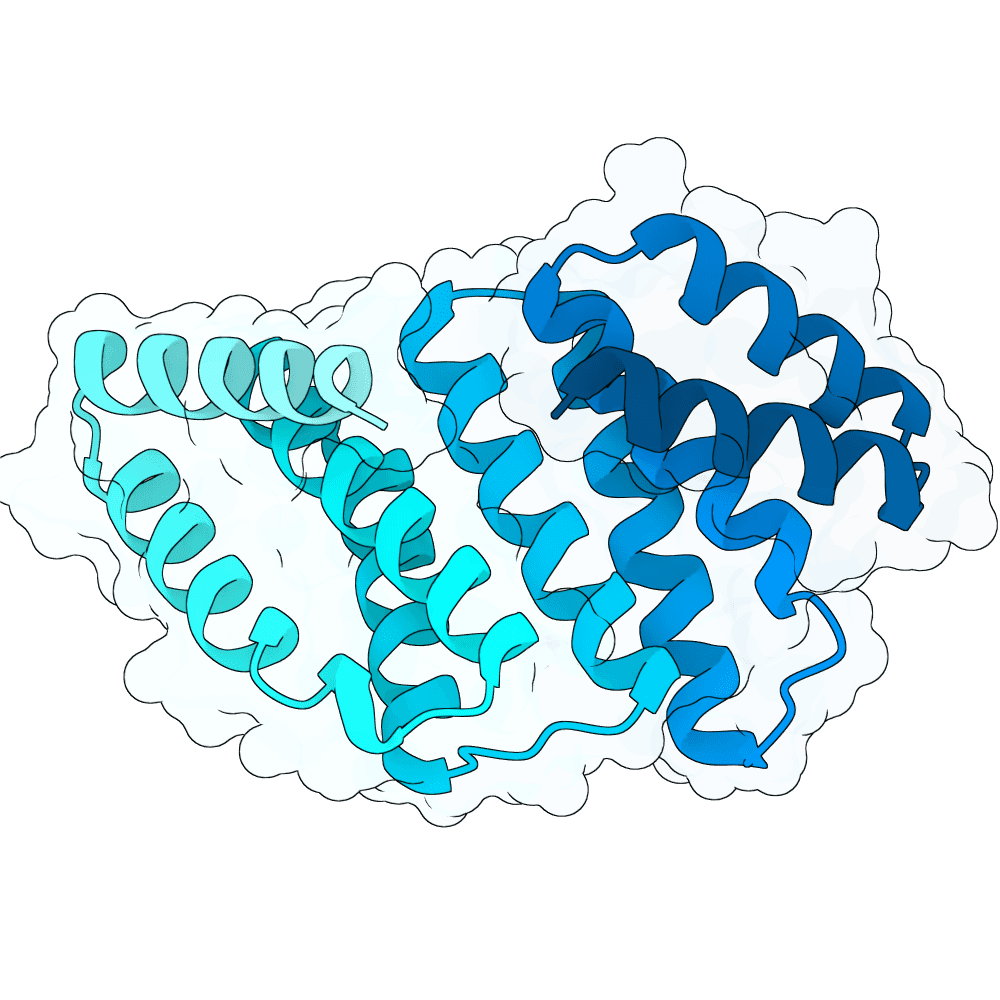
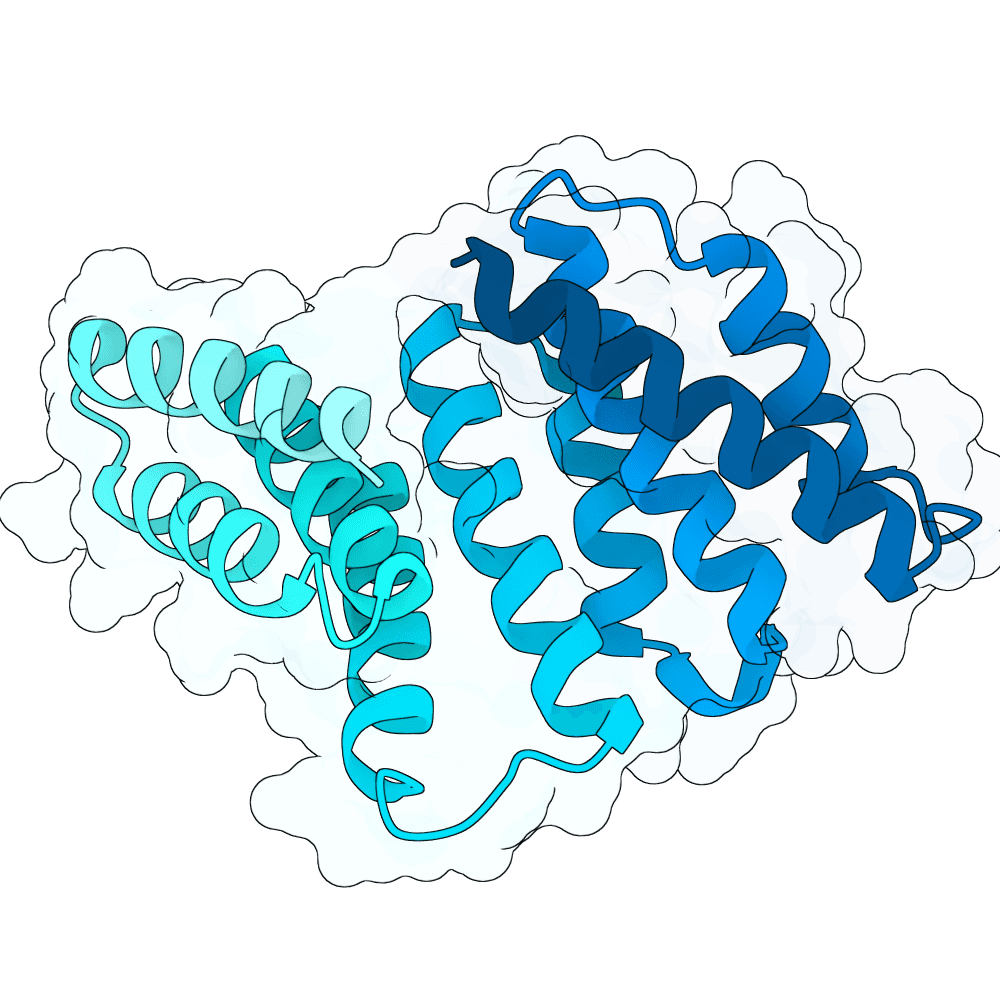
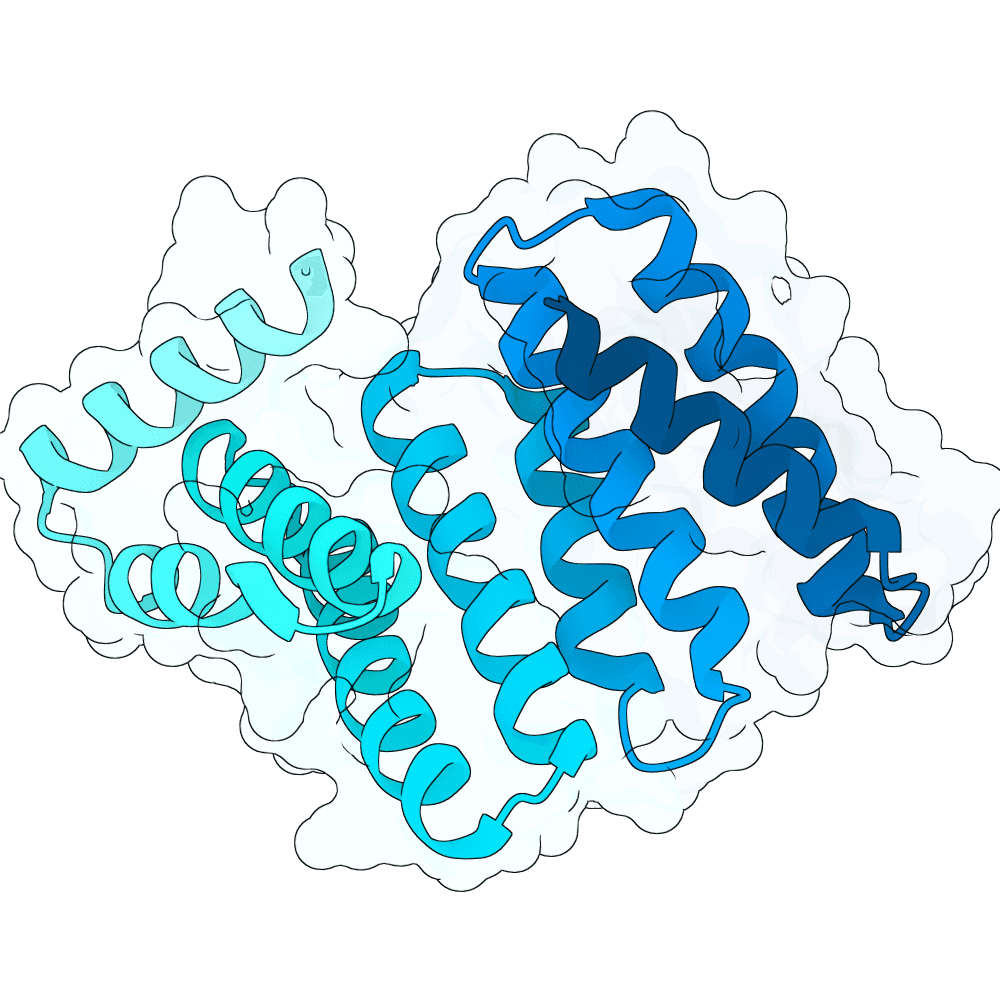
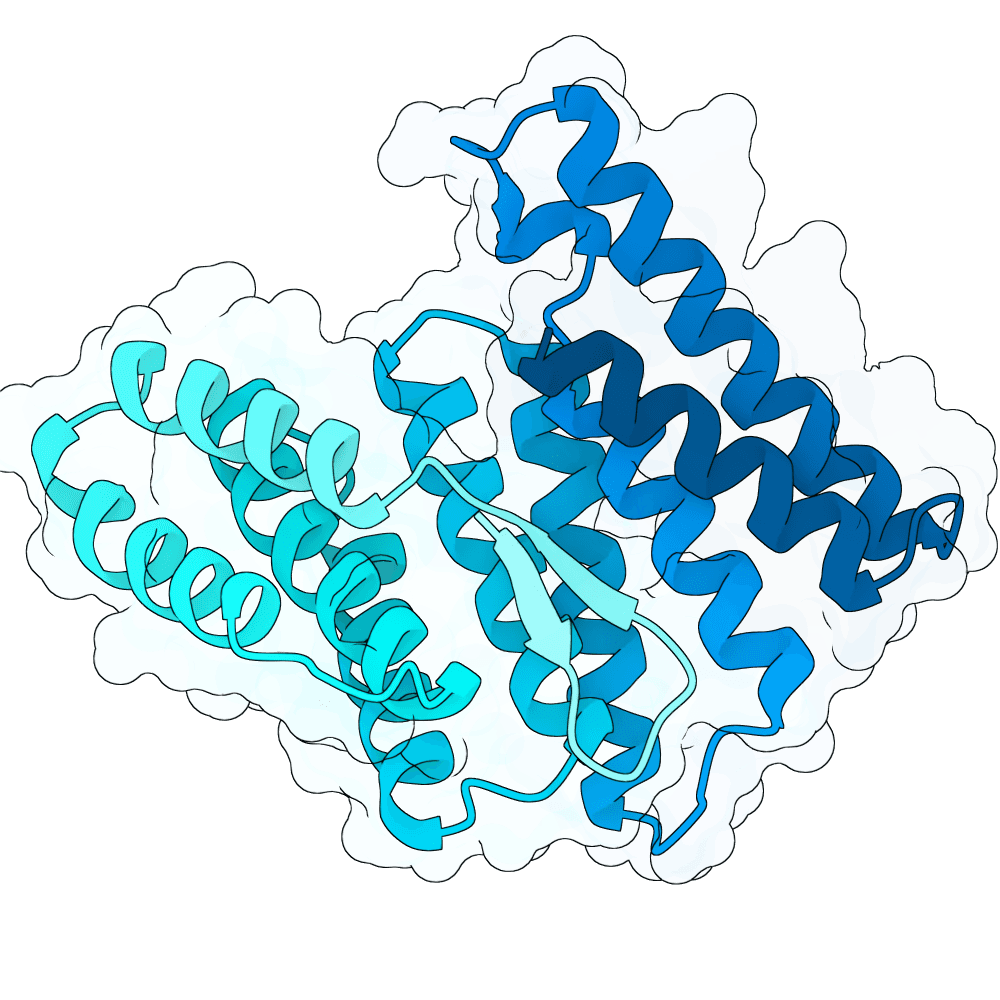
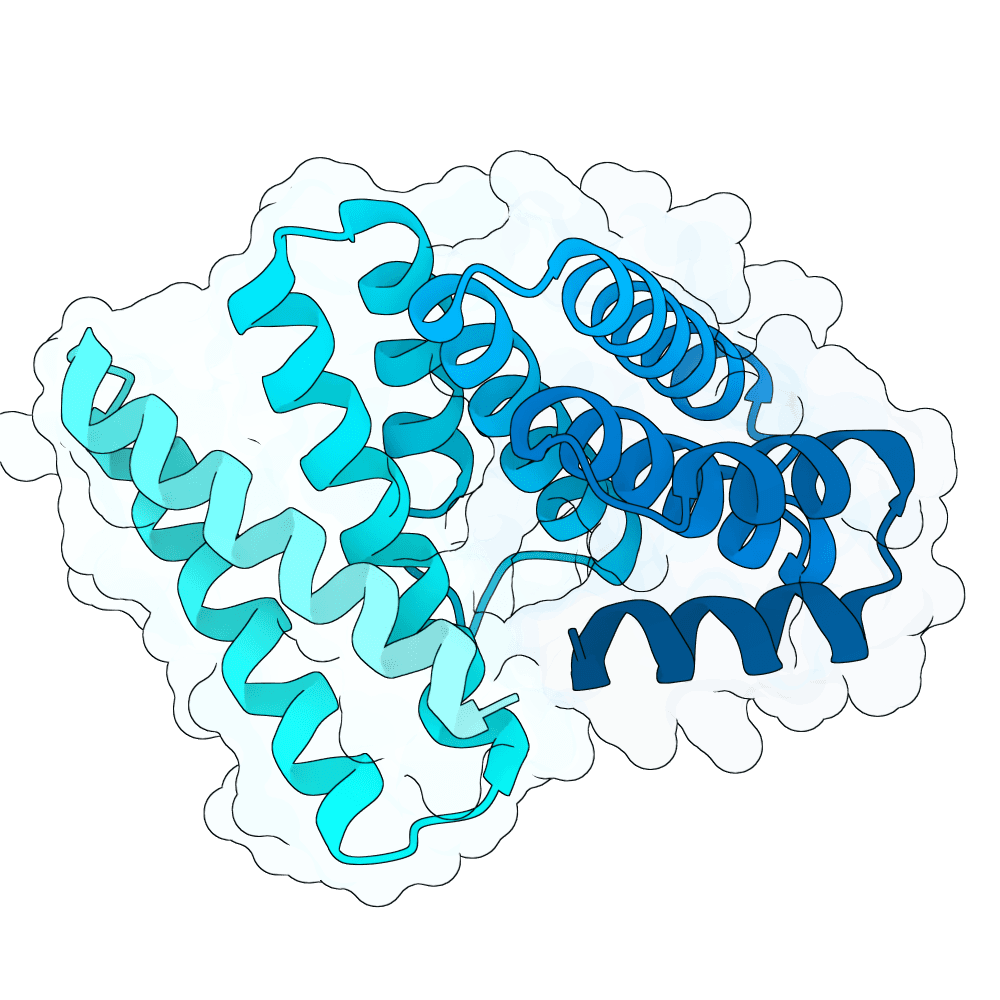
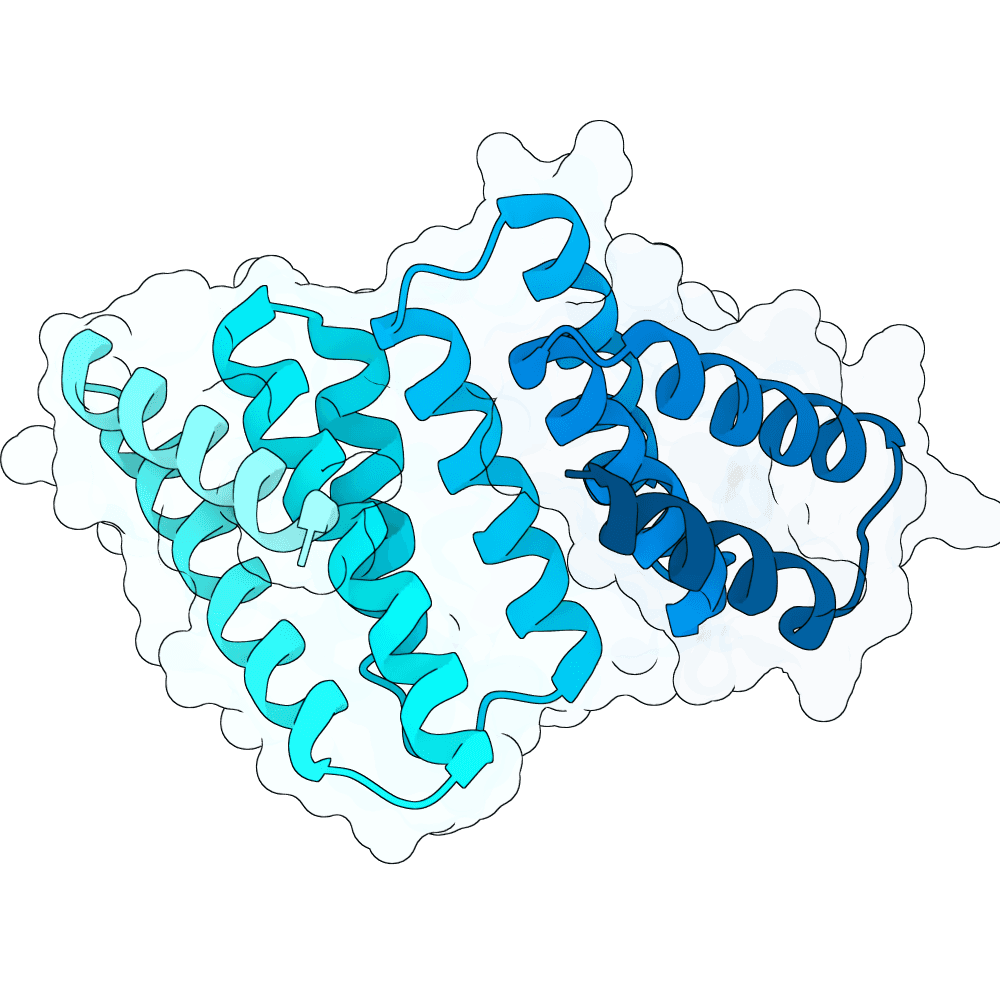
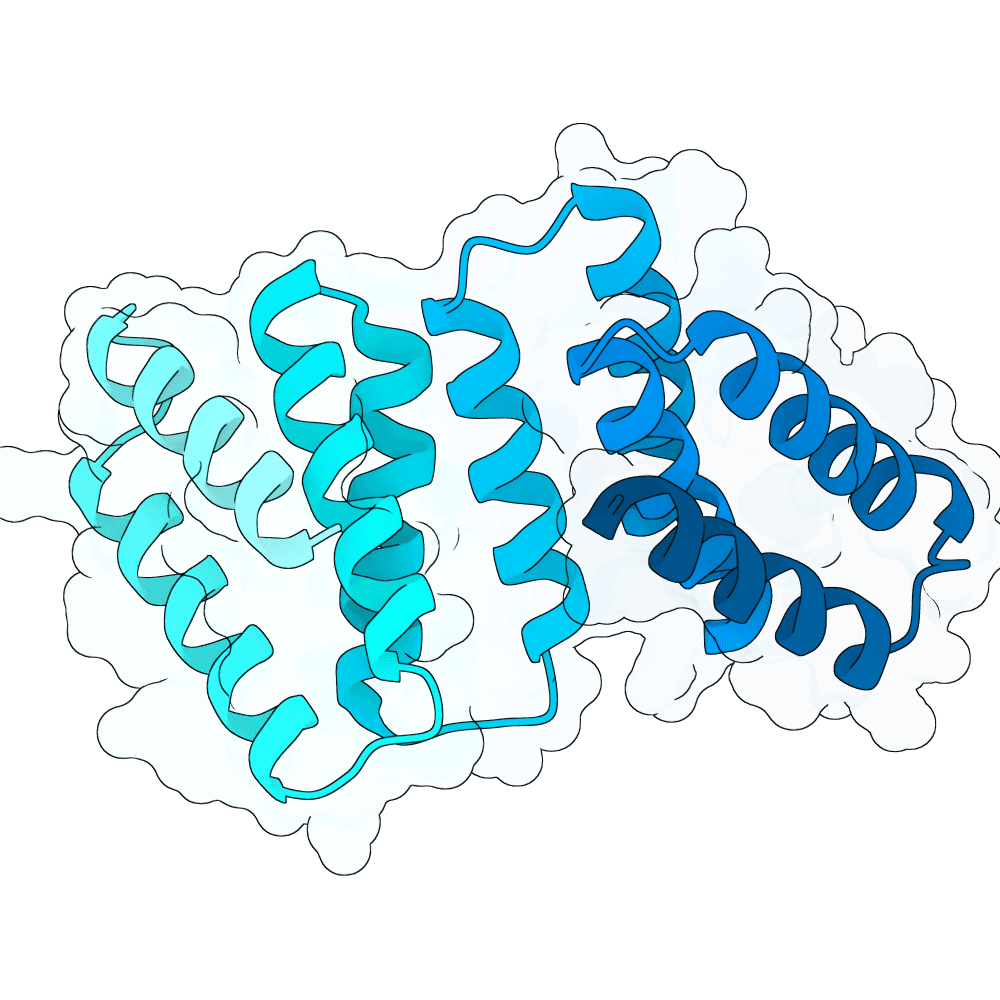
For all our designs we used the crystal structure of the Glomulin-RBX1-CUL1 complex (PDB: 4F52). We decided to try a number of different approaches to design binders against RBX1 and ultimately group them by target area / seed and choose the sequences with the best ipSAE. We generated binder designs using the following methods:
- Boltz-Lab & TIMED-Charge
Using Boltz-Lab (https://lab.boltz.bio/), we generated ~5K designs targeting two main regions of the RBX1: the unstructured region (“tail”), and the structured region (“head”). For the tail, we used a completely de novo design generation, while for the head, we partially diffused the helical bundle of the Glomulin binder from structure PDB: 4F52. We scored the designs with Boltz-Lab's binding confidence and manually filtered down by diversity of fold to 18 “seed” designs: 15 for the tail and 3 for the head.
We then used TIMED-Charge (https://doi.org/10.1093/protein/gzae002) for sequence design. We systematically mutated residues with lower predicted probability towards higher probability for up to 5 positions for the tail, and until sequence similarity decreased below the 25% threshold for the head. Due to a bug in Boltz-Lab we also redesigned UNK residues. We generated a total of ~17K sequences.
Finally we selected 450 sequences for folding using Boltz-2: a consensus sequence for each of the 18 seeds, the top scoring sequences by ESM PLL, and 25 random sequences for good luck.
- Boltz-lab & TIMED-Charge + Mosaic Optimisation
For each of the seed designs, we created a Position Weight Matrix (PWM) with all the designed sequences. Then we modified Mosaic to accept the PWM to optimise the designs by iPSAE and skip the random sampling. We used Mosaic on Modal to optimise 3 tail designs, generating a total of 50 designs.
- Re-implementation of Wu et al. without pyRosetta
Binder design followed the general workflow described by Wu et al. (https://doi.org/10.1126/science.adr8063) and was executed using a portable Nextflow (DSL2) reimplementation of the original pipeline. The overall strategy: template-based peptide threading, ProteinMPNN sequence design, structure prediction, confidence-based filtering was preserved. Three components differ from the original publication. First, peptide threading was performed using a lightweight Python module employing BLOSUM62 substitution scoring and a sliding-window approach, replacing the physics-based PyRosetta FlexPepDockingProtocol used in the original work. Target peptide windows are slid across template peptide positions and filtered by minimum BLOSUM62 score (threshold: −10) and sequence identity; only backbone atoms are retained in the threaded output structures. Second, structure prediction for filtering was carried out with Boltz2 (https://doi.org/10.1101/2024.11.19.624167) instead of AlphaFold2-multimer with an initial-guess seed as described in the original protocol. Designed sequences were submitted to Boltz2 and the resulting models were filtered by ipTM ≥ 0.83, pLDDT ≥ 85, and interface RMSD ≤ 2.0 Å. Third, the pipeline runs as a single linear pass without the iterative MPNN→structure-prediction refinement rounds or optional RFdiffusion scaffold diversification described in the original work. Sequence design itself was performed with ProteinMPNN, identical to the original implementation.
- Mosaic without inverse folding and ranking
We used the winning approach for de novo binder design from the recent Nipah Binder competition (https://proteinbase.com/competitions/adaptyv-nipah-competition) of using Mosaic for binder hallucination and skipping inverse folding and filtering (https://blog.escalante.bio/180-lines-of-code-to-win-the-in-silico-portion-of-the-adaptyv-nipah-binding-competition/). Due to limited budget Modal, we only produced 5 designs this way.