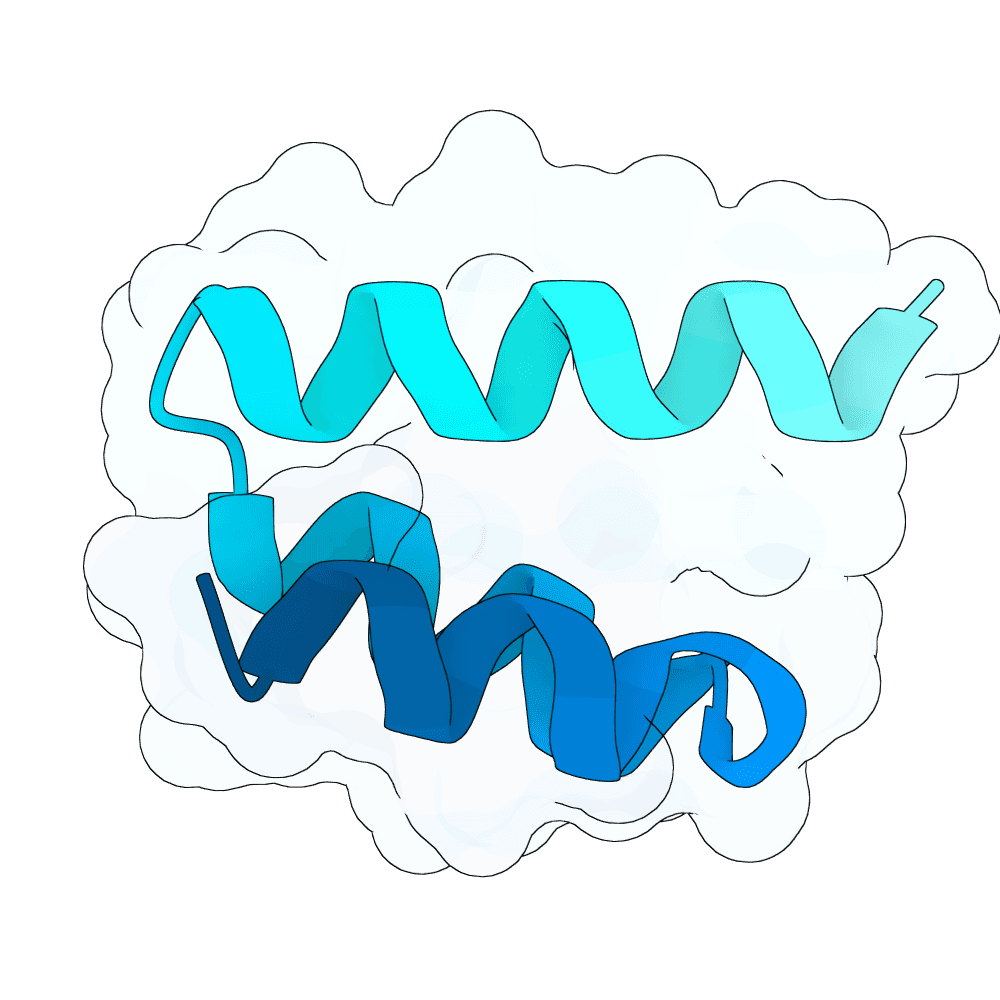Description
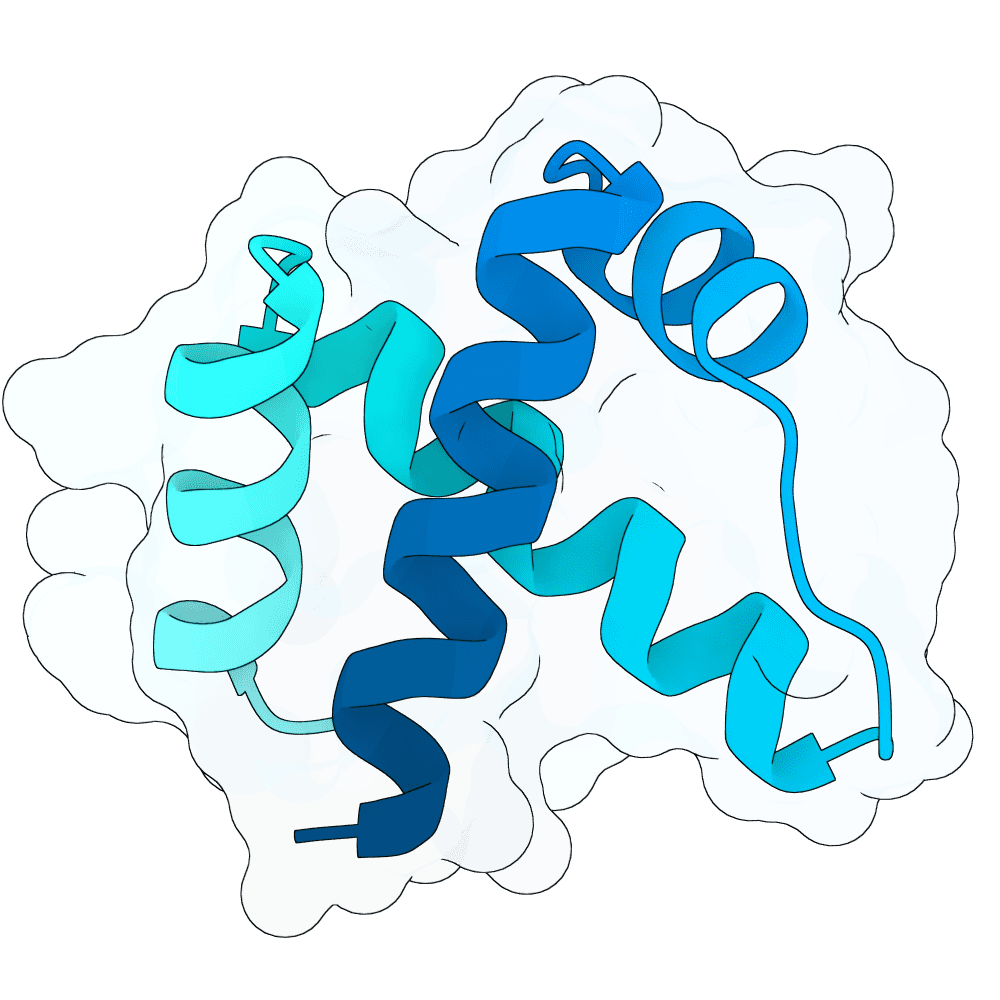
This project focuses on de novo design of compact protein binders targeting the RBX1 RING domain. The working hypothesis was that the RBX1-Glomulin interface captured in PDB 4F52 represents a structurally validated proxy for the functional E2-binding surface of RBX1. Because Glomulin is a natural RBX1 inhibitor that occupies this region, we reasoned that a designed binder engaging the same epitope could competitively block E2 recruitment and thereby interfere with SCF ubiquitin ligase function.
To define the design epitope, we started from the 4F52 structure and performed residue-level analysis of the RBX1-Glomulin interface. This identified a set of hotspot residues on RBX1, including Asn47, His48, Ile54, Glu55, Gln57, Ala58, Arg86, and especially Trp87, which forms a dominant hydrophobic contact in the native inhibitory complex. These interface features were then mapped onto the RBX1 NMR structure 2LGV and encoded into the design specification file as explicit binding hotspots. At the same time, the flexible N-terminal region was excluded from productive binding and hidden from the generative model so that sampling was focused on the structured RING epitope rather than non-functional contacts.
A critical design constraint is the presence of coordinated Zn2+ ions, which are required to maintain the RBX1 RING fold. Ignoring the metal would risk local distortion of the target structure and introduce artificial clashes between designed binders and the coordination shell. For that reason, all design stages were carried out in an all-atom setting that explicitly retained the Zn2+ ions and their local structural context. Using this setup, an all-atom binder design model boltzgen was applied to generate 10,000 candidate complexes, which were then filtered and ranked based on hotspot engagement, interface placement, steric compatibility, and overall structural quality. The top 100 candidates were retained for more detailed analysis.
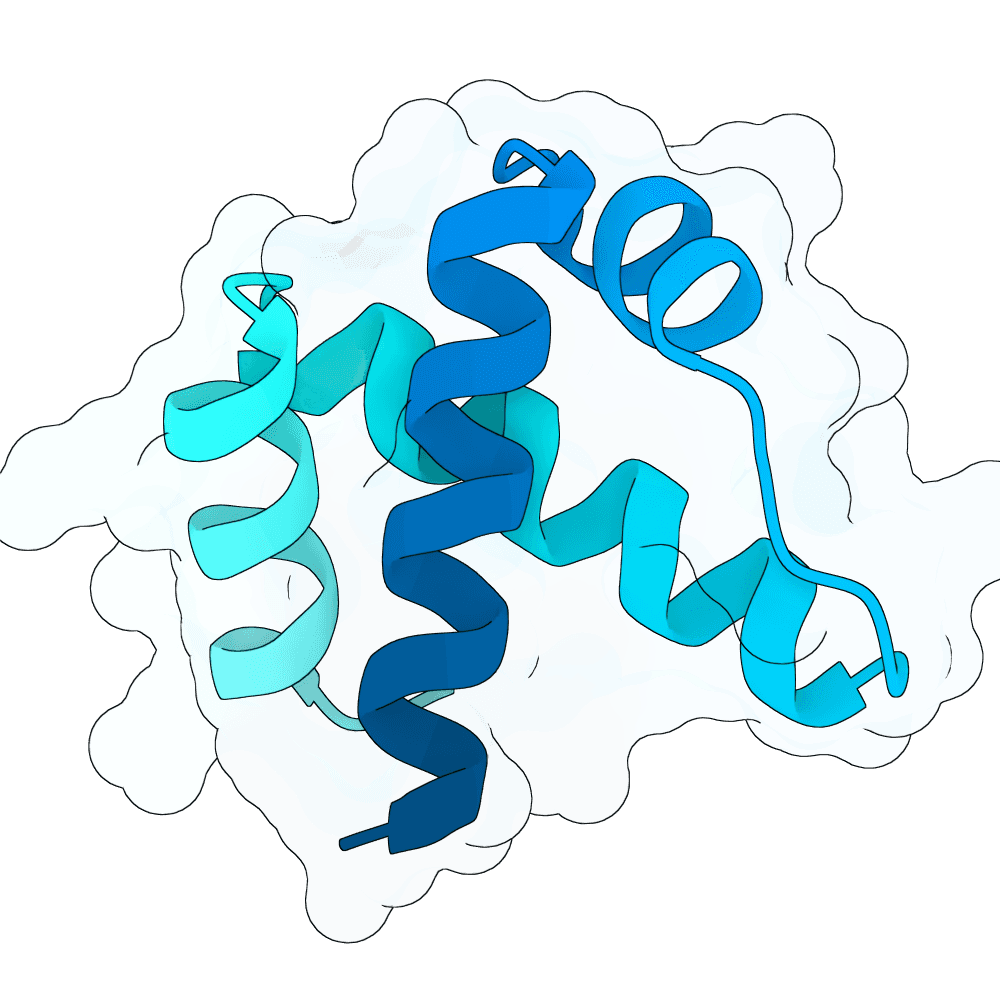
The next stage focused on identifying the most robust scaffolds rather than simply the highest-scoring single models. The top 100 candidates were evaluated using binding-site contacts, total interface contacts, a buried-interface proxy, and structural compatibility with the RBX1 RING conformation in 2LGV. Because 2LGV is an NMR ensemble, we also examined compatibility across multiple conformations rather than relying on a single target model. The RBX1 epitope proved to be highly rigid across the ensemble, supporting the expectation that designs optimized against one representative model should remain compatible with the broader solution-state conformational space. Based on these criteria, ten preferred scaffolds were selected. These candidates represented the best balance among hotspot coverage, epitope matching, interface quality, and conformational robustness.
Sequence optimization for the selected scaffolds was then performed with LigandMPNN in atom-context mode so that both the target environment and the Zn2+-dependent local geometry were considered during redesign of the binder chain. This step was intended to improve sequence-structure compatibility while avoiding sequences that would be incompatible with the metal-stabilized target surface. Multiple sequences were generated for each scaffold under conservative sampling conditions.
Finally, the redesigned binder-target complexes were re-evaluated using AF2 initial guess. The purpose of this step was to test whether the designed sequences could refold back to the intended complex geometry while preserving the designed interface. In other words, only candidates that remained self-consistent after an independent structure prediction step were prioritized. The overall strategy was therefore to enforce agreement across four levels: biologically informed epitope definition from 4F52, metal-aware all-atom backbone generation, target-context sequence redesign, and structural recovery by AlphaFold2-based validation. The outcome is a focused set of RBX1 binder candidates prioritized for downstream experimental testing rather than a claim of experimentally confirmed binding.